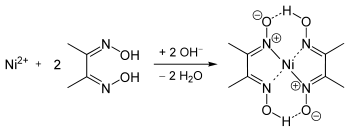
Ni Dmg 2 Molecular Weight Loss
Ni(dmgH) 2 complex can serve as active sites to promote H 2 generation of MOFs. The efficient charge transfer from MOF to Ni(dmgH) 2 causes the enhanced H 2 production. The low cost and easy operation makes Ni(dmgH) 2 having great potential for photocatalytic H 2 production. ›› Nickel molecular weight. Molar mass of Ni = 58.6934 g/mol. Convert grams Nickel to moles or moles Nickel to grams ›› Percent composition by element. Its abbreviation is dmgH 2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the dioxime derivative of the diketone butane-2,3-dione (also known as diacetyl). DmgH 2 is used in the analysis of palladium or nickel. Lion 10.7 dmg download tpb. Its coordination complexes are of theoretical interest as models for enzymes and as.
- Synonym: Ni(dppf)Cl 2. CAS Number 67292-34-6. Empirical Formula (Hill Notation) C 34 H 28 Cl 2 FeNiP 2. Molecular Weight 683.98. MDL number MFCD00270284.
- 288.7 g of Ni (DMG)2 = 58.7 g of Nickel. So 'x’ g of Ni (DMG )2 = y. By simplifying the above relation you can estimate the amount of nickel present in the given solution by gravimetric method with the help of DMG. Thanks for asking! Hope it helps!
| Physical Chemistry Virtual Lab Physical chemistry (also called physicochemistry) is the explanation of macroscopic, microscopic, atomic, subatomic, and particulate phenomena in chemical systems in terms of physical concepts; sometimes using the principles, practices and concepts of physics like thermodynamics, quantum chemistry, statistical mechanics and dynamics. Spectrophotometry || Cryoscopy || Ebullioscopy || EMF measurement || Determination of Viscosity of Organic Solvents || Adsorption Isotherm || Verification of Tafel Equation || Determination of Viscosity Average Molecular Weight of Polymer || Calorimetry -Water equivalent Calorimetry || Calorimetry -Heat of Neutralization |
| Organic Chemistry Virtual Lab Organic chemistry is a discipline within chemistry which involves the scientific study of the structure, properties, composition, reactions, and preparation (by synthesis or by other means) of chemical compounds that contain carbon. Detection of Functional Groups || Detection of Elements: Lassaigne鈥檚 Test || Separation of Compounds Using Column Chromatography || Purification by Fractional distillation/crystallisation || Purification by Steam distillation/crystallisation || Laser Flash Photometer || Organic Preparations - Allylation of Isatin || Estimation of Aspirin || Estimation Of Glucose || Calculation of 位max of Organic Compounds Using Woodward Fieser Rules |
| Inorganic Chemistry Virtual Lab Inorganic chemistry is the branch of chemistry concerned with the properties and behavior of inorganic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds). Water analysis-Determination of Physical parameters || Water analysis-Determination of Chemical parameters || Acid Base Titration || Gravimetric Estimation of Barium || Gravimetric Estimation of Nickel || Crystal Field Theory || Group Theory || Alloy Analysis (Brass) || Soil Analysis-Determination of Specific conductivity of Soil || Soil Analysis-Determination of pH of Soil |
| Advanced Analytical Chemistry Virtual Lab Analytical chemistry is the branch of chemistry concerned with studying the properties of materials and development of tools used to analyze materials. It is the science of sampling, defining, isolating , concentrating and preserving samples. Soil Analysis-Determination of Available Organic Carbon content in the Soil || Soil Analysis-Determination of Available Nitrogen content in the Soil by Kjeldahl method || Soil Analysis-Determination of Available Phosphorus content in the Soil by Bray's method || Electrogravimetric Estimation of Metals || Estimation of Phosphate Content in Soft Drinks || Flame Photometry || Polarography - Determination of Unknown Concentration of Cadmium || Polarography - Determination of Unknown Concentration of Vitamin C |
